Electron orbitals
= the place where an electron is normally found as it spins around the nucleus. See Figure 9 in your textbook. Each orbital possesses a certain amount of energy due to its location (distance) from the nucleus. In other words, we say that each electron orbital possesses an energy level or electron shell.
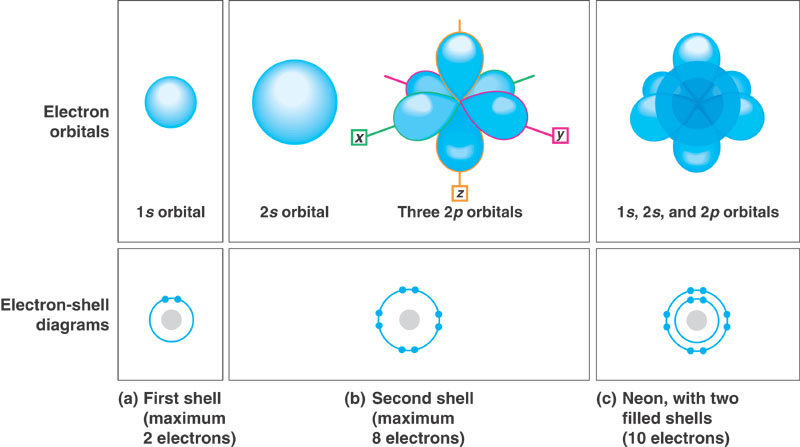
For the sake of simplicity, we will not discuss the specific orbitals (e.g., s and p orbitals) but rather will just discuss the concept of electron shells.
Atoms chemically react with other atoms in an attempt to fill the outermost shell. The second and third shells contain a maximum of eight electrons (octet).